|
This patent is for sale. Request on: info@rue25.de
Referenz: DPMA Nr: DE 10 214 013 701.5
Filed Date: 2014-09-28
Label: MagFieldCat_2014
A method using a catalyst which is amplified by a magnetic field
Description
The present invention describes a catalyst which is influenced by a changing magnetic field. The magnetic particles of a catalyst are mixed by an alternating magnetic field in motion, thereby increasing the interfacial contacts of different particles. This allows molecules, that activate themselves by physical and chemical adsorption on the surfaces and adhere as an intermediate molecule on the surface, to be adopted by other catalytic groups. In addition it creates an attraction and rejection between the catalyst and the reacting molecules, by varying the displacement of the electric charge carriers.
By this invention, a higher conversion of the catalytic reaction takes place.
STATE OF THE ART
Catalysts often consist of a mixture of several chemical components, which show the property in this combination to convert an original material (educt) into a new specific material (product) by using the chemical and physical adsorption to
by create an intermediate product, which in a subsequent step is converted again.
Studies have shown that a higher yield is achieved in the targeted conversion, when a plurality of different chemical elements, together act as a catalyst.
Thus, for example, in recent studies, the mixture of nickel plus gallium NiGa or Ni5Ga3 is used for the conversion of carbon dioxide and hydrogen to methanol.
A similar property has the mixture of copper plus zinc oxide plus alumina oxide.
The mixtures can be composed of an alloy or as an simple mixture of the elements.
The technical implementation of these mixtures are given either as a mixed powder or a sintered material in a reactor, which is then filled with a reactive gas mixture or liquid mixture. It is then converted by means of the catalyst under temperature and pressure. The reactor embodiments can be configured as a flow-through reactor or as fill-reactors which are cyclically filled and emptied, and are often technically stirred during the reaction time.
Most chemical-mechanical processes are not elucidated in detail, but it turns out that the reactions are highlighted mainly on special interfaces. So it is quite relevant, how the microscopic surfaces are shaped . Crystalline forms and shapes, such as those caused by gallium, can mean a substantial increase.
Disadvantage of the current situation
The disadvantage of the shown mixtures is, that the surfaces behave like independent local regions. This results in many border areas which create chemical adsorption and intermediate molecules in a first step, but these molecules are only insufficiently or not converted into subsequent processes, since the real distribution of chemical constituents are not distributed equally.
Regions with dominance of single chemical catalyst element, can not be fully active.
TASK
The invention shall increase the catalytic turnover of molecules by increasing the contacting of different interfaces and mixing them.
SOLUTION
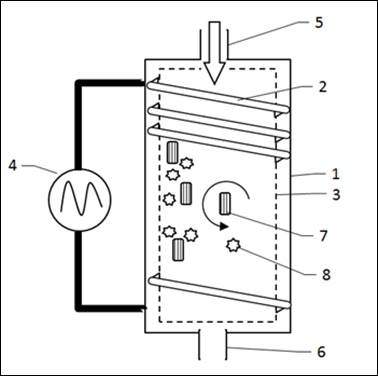
(Fig.1): In a reactor (1) is an inner catalytic chamber (3).
The inner catalyst chamber is surrounded by a electic coil (2). The electrical coil produces a magnetic field which flows through the catalyst compartment as soon as the current flows through the coil.
Fig. 1
When there is a change of the electric current in the coil in direction and or magnitude, then the magnetic field changes in the catalyst space.
(Fig. 2) By changing the magnetic field which passes through the catalyst, the magnetically active particles (7) moves, thereby the adsorbinging interfaces of the various components touch alternately.
Fig. 2
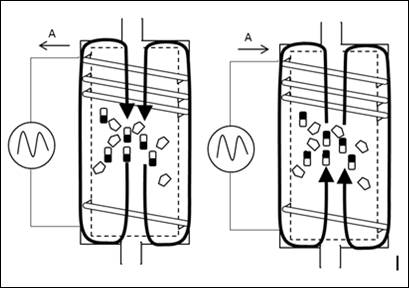
Thereby it is achieved that the intermediate products are contacted to the surfaces of the catalyst particles from the second or third catalyst elements and hence secondary reactions can be better realized.
It will also find contacts with magnetically inactive particles (8).
The rotation or movement of the magnetic active particles thus acts like a mill on a microscopic size.
The reactor (1) is designed at the approach as a flow reactor with an input opening (5) and an output (6). The reaction can also be scheduled for one-sided reactors, which are only filled and emptied by one page.
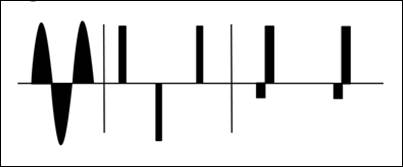
(Fig.3) The optimum excitation of the particles can be controlled with various current forms. First, of course, an AC signal in sinusoidal or rectangular shape is possible. The frequency and amplitude can be designed differently according to as needed. Also it has not to be only a symmetrical design of the excitation current and thus the magnetic field. It may also the target to change the particles only partially similar to an occasional shaking.
Fig. 3
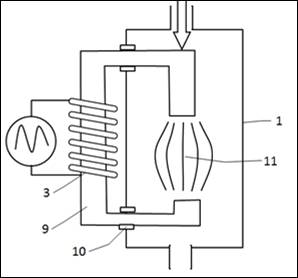
(Fig.4) The magnetic excitation system can be built externally and be passed over a magnetic core into the reactor. Therefore the coil (3) has to be established on a magnetic core (9). The magnetic core is then fed via insulators (10) into the reactor and in the reactor, the magnetic core is then interrupted. Inside the interruption (11) the magnetic field expands and flows through the catalyst.
Fig. 4
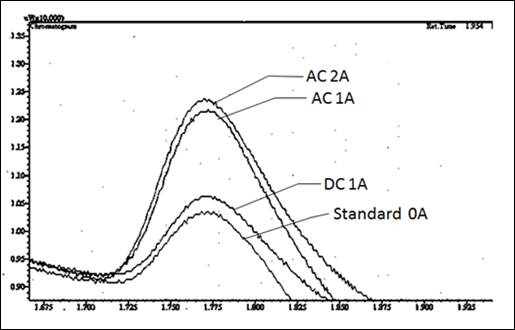
(Fig.5) As an example the effect, the reactions are illustrated with reference to a structure, where the catalyst consisted of nickel and gallium oxide and gallium. This was led to the synthesis of methanol from carbon and hydrogen
The reactor was passed through a pipe reactor with a gas mixture of carbon dioxide and hydrogen. The reactor was heated to about 400 degrees and maintained, the gas having a pressure of 20 bar.
Since nickel is an magnetically reactive element, it aligns to the magnetic field, and so the catalyst could be energized during operation of the coil.
The measured results were made on a gas chromatography of the type Shimadzu GC 14A-B and show that more methanol is formed, when the coil with an AC current of 50 Hz and 1.5 amps and 2.5 amps are activated. The excitation with a direct current DC did not improve.
Fig. 5
The drawings show the following representations:
Figure 1: Overall structure of a reactor
Figure 2: Changing magnetic field in reactor
Figure 3: Possible characteristics of the excitation current
Figure 4: External excitation
Figure 5: Example of increase in the production of methanol
List of designations in the drawings
1 reactor
2 coil
3 Inner catalyst-room
4 Power Source
5 Input
6 Output
7 Electromagnetic effective catalyst particles
8 Other catalyst particles
9 Magnetic core
10 Insulation
11 Interruption of the magnetic core
|
|
|
|
.
|